If there were only one number to gauge your overall health, fitness, and longevity, it would be...
Concussion Treatment - HBOT's Efficacy Under Pressure
On July 6, 2015, we wrote to the New York Times about an article that appeared on the front page of the July 3rd Business Day section, titled “Effective Concussion Treatment Remains Frustratingly Elusive, Despite a Booming Industry.” This letter has been modified and expanded below.
We were thrilled to see the article by Barry Meier and Danielle Ivory in the New York Times discussing potential alternative treatments for concussions, or mild traumatic brain injuries (mTBI), including the use of HBOT. Nevertheless, we were disappointed with the short-shrift given to HBOT by many of the physicians quoted in the article.
Along with our colleagues in the field of HBOT—including Dr. Harch, featured in the article—we have witnessed great improvements through the use of HBOT in the treatment of mTBI patients. Many of our patients have experienced profound life-changing results, including reduced irritability and fatigue, improved concentration and memory, and less headaches and dizziness.
The Defense Department has spent more than $800 million on brain injury research.
Credit: Zach Gibson for The New York Times
It is important to clear some things up. The studies cited in the article as showing HBOT’s lack of effectiveness have major flaws which should not be overlooked. The most important is that the “sham,” or placebo groups, in virtually all of the VA-sponsored studies were also treated in a hyperbaric chamber, but were given less pressure and/or less oxygen than that of the treatment group. Oxygen saturation in the blood in these supposed “sham groups,” however, was increased, on average, by about 50%. As a result, the findings did not reflect true placebo-controlled trials.
You see, in the process of HBOT, as soon as the chambers are turned on, pressurization begins and physiology changes, no matter if the chamber is pressurized with oxygen, pressurized with air, or nitrogen levels are increased instead. There are countless studies that reflect this to be the case. The fact of the matter is that all patients treated in these VA-sponsored studies who had an increased blood oxygen concentration in the body experienced some degree of improvement.
An alternative study design is a randomized crossover control study, where subjects are randomized to two groups; both groups are treated with the therapy in question, just at different points in time. This study design allows for what is known as the “triple comparison”—between treatments in the two groups, between treatment and no treatment in the same group, and between treatment and no treatment in different groups. This study design still has its challenges, but the fact of that matter is that there is no way to conduct a true randomized prospective placebo-controlled hyperbaric study unless everyone in the trial has tympanic membrane tubes or incisions, so neither the placebo nor the treatment group could feel pressure. Such a trial design would likely lead to ethical concerns, given the invasive nature of the procedure; although it should be noted that, in adults, tympanic tubes or incisions can be done in an office setting without any sedation and minimal long-term risk.
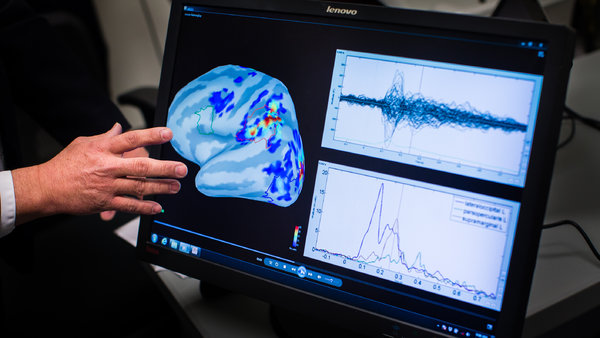
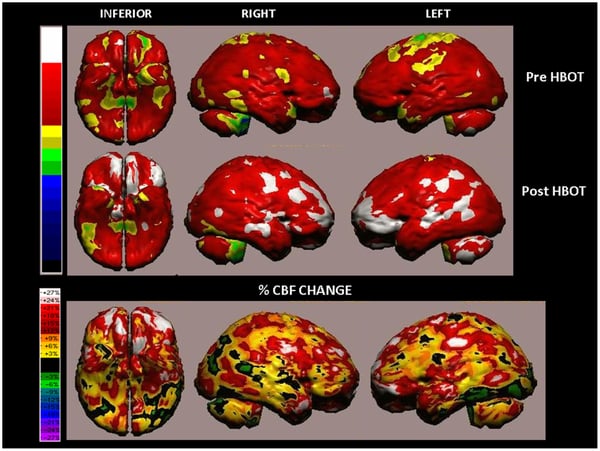
Dr. Shai Efrati and his team at Tel-Aviv University have embraced the crossover trial design as the best way to study the effects of HBOT and have published some very innovative research on the use of HBOT for mTBI (they also have published a stroke study as well). In his study titled “Hyperbaric Oxygen Therapy Can Improve Post-Concussion Syndrome Years after Mild Traumatic Brain Injury – Randomized Prospective Trial,” Dr. Efrati and colleagues studied 56 mTBI patients using SPECT imaging, cognitive evaluations, and quality of life testing, and concluded that HBOT can induce neuroplasticity— the brain’s ability to form new neural pathways—leading to the repair of chronically impaired brain functions and improved quality of life in mTBI patients. SPECT scans show the brain in real-time and measure blood flow. Dr. Efrati and colleagues used SPECT scans to measure the changes in blood flow after HBOT; these changes were not only improved dramatically, and with statistical significance, but also correlated to improvements on a battery of objective cognitive tests.
In the end it's about helping patients as safely and as effectively as we can. Above all, we know that HBOT is safe and minimizes the harsh side effects off-label pharmaceuticals traditionally used to treat mTBI patients can cause. Across the country, we are having great results using HBOT as part of a multidisciplinary approach to improve the lives of mTBI patients—both veteran and civilian. Pharmaceuticals are often used to treat mTBI, but these are just short-term patches, and do not actually treat the underlying reason for the mTBI: a chronic non-healing wound in the brain. HBOT heals wounds, no matter where they are, and we are seeing this now on SPECT and specialized MRI scanning images before and after HBOT sessions.
HBOT is not a “snake oil” used in the treatment of concussions. While it is certainly not a matter of “one size fits all,” many of our patients, and those of our colleagues around the country, have experienced profound results using HBOT.
Written by Dr. Henry Prince and Dr. Scott Sherr
...